 
By measuring the deflection and knowing the strength of the electric and magnetic fields, Thomson was able to calculate the charge-to-mass ratio of the electron: $\boldsymbol$. He observed that the rays were deflected by these fields, indicating that they were charged particles. Thomson's experiment involved applying an electric field and a magnetic field perpendicular to the path of the cathode rays. He discovered that these rays were composed of negatively charged particles, which he called "corpuscles," later known as electrons. Thomson conducted a series of experiments using cathode ray tubes to investigate the properties of cathode rays. Thomson's Cathode Ray Tube Experiment and Atom Model: Cathode rays or streams of electron particles are quite easy. Thomson, led to the discovery of the negatively charged part of the atom, the electron. The function of the cathode ray tube is to convert an electrical signal into a visual display. Thomson's model of the atom was shown to be incorrect in Geiger-Marsden's gold foil experiment.J.J. The cathode ray tube experiment, originally carried out by J.J. 
the smallest particle of an element that retains its identity in a chemical reaction. In chemical reactions, atoms are combined, separated, or rearranged. This feature of the model was based on theory and lacked experimental evidence. Atoms of different elements combine in simple whole-number ratios to form chemical compounds. One major limitation of Thomson's model is the lack of explanation for the positive mass. The charge to mass ratio of an electron was determined from Thomsons cathode-ray tube experiment to be 1.759 × 108 C/g and the charge on a single electron was determined from the Millikan oil drop experiment to be 1.602 × 10-19 C, so the mass of a single electron is. The electrons are held together in the atom due to their attraction to the positive mass. In Thomson's model of the atom, negatively charged electrons are dispersed in a positive mass due to electrostatic repulsion. The discovery of the electron as a subatomic particle led to the development of Thomson's atomic model.Ī simple representation of Thomson's atomic model Thomson's Model of the Atom ('Plum Pudding' Model) Thomson assumed the magnitude of charge is equal for these particles, leading to the conclusion that the mass of an electron is 1800 times smaller than a hydrogen ion. The charge to mass ratio of electrons was shown to be 1800 times greater than that of hydrogen ions. Thomson and essentially discovered the existence of electrons. As a result, Thomson's experiment led to the discovery of the first subatomic particle – the electron. The Cathode Ray Experiment was performed by J.J. In addition, the constancy of the cathode ray's charge to mass ratio shows that these negatively charged particles are present in all matter. Since an actual charge to mass ratio value was determined for cathode rays, Thomson proved that cathode rays do indeed have mass, and hence are negatively charged particles. He showed that the value of charge to mass ratio remained constant. Thomson replicated the experiment using cathodes made from different metals and under various conditions. Since the value of E, B and r were known to Thomson, the value of the charge to mass ratio of the cathode ray was calculated. (We recommend understanding the derivation of the above equation to be able to provide mathematical support in exam responses) 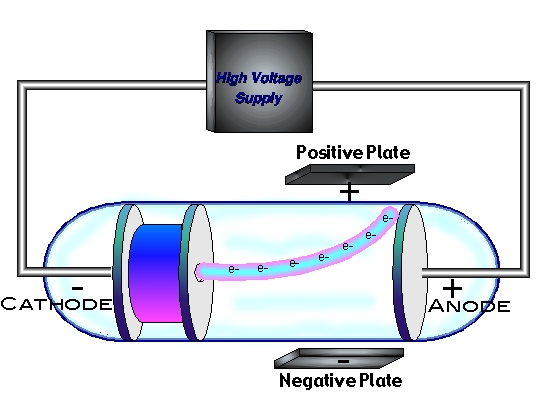
The centripetal force was provided by the force due to the magnetic field: Here, the radius of the cathode's curved path can be analysed by considering centripetal force. The results of these experiments helped Thomson determine the mass-to-charge ratio of the cathode ray particles, which led to a fascinating discovery - minus the mass of each particle was much, much smaller than that of any known atom. Under the influence of the magnetic field only, the cathode ray was deflected downwards. Thomson also placed two magnets on either side of the tube, and observed that this magnetic field also deflected the cathode ray. When this occurred, the force acting on the cathode ray due to the electric field balanced the force due to the magnetic field.Īfter a straight trajectory was achieved, the electric field was switched off. Thomson adjusted the strength of the magnetic field until the cathode travelled a straight path. The direction of this magnetic field was perpendicular to the electric field, and positioned such that it would cause the cathode ray to deflect downwards. Next, Thomson applied a uniform magnetic field using current-carrying coils. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
